Home » Health News »
Unlocking the secrets of the brain’s dopaminergic system
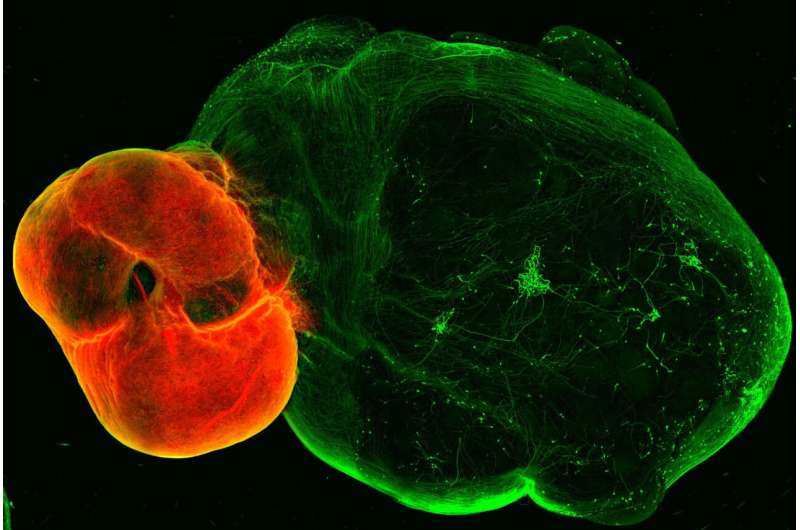
A new organoid model of the dopaminergic system sheds lights on its intricate functionality and potential implications for Parkinson’s disease. The model, developed by the group of Jürgen Knoblich at the Institute of Molecular Biotechnology (IMBA) of the Austrian Academy of Sciences, replicates the dopaminergic system’s structure, connectivity, and functionality.
The study, published in Nature Methods, also uncovers the enduring effects of chronic cocaine exposure on the dopaminergic circuit, even after withdrawal.
A completed run, the early morning hit of caffeine, the smell of cookies in the oven—these rewarding moments are all due to a hit of the neurotransmitter dopamine, released by neurons in a neural network in our brain, called the “dopaminergic reward pathway.”
Apart from mediating the feeling of “reward,” dopaminergic neurons also play a crucial role in fine motor control, which is lost in diseases such as Parkinson’s disease. Despite dopamine’s importance, key features of the system are not yet understood, and no cure for Parkinson’s disease exists. In their new study, the group of Jürgen Knoblich at IMBA developed an organoid model of the dopaminergic system, which recapitulates not only the system’s morphology and nerve projections but also its functionality.
A model of Parkinson’s disease
Tremor and a loss of motor control are characteristic symptoms of Parkinson’s disease and are due to a loss of neurons that release the neurotransmitter dopamine, called dopaminergic neurons. When dopaminergic neurons die, fine motor control is lost, and patients develop tremors and uncontrollable movements. Although the loss of dopaminergic neurons is crucial in the development of Parkinson’s disease, the mechanisms of how this happens and how we can prevent—or even repair—the dopaminergic system are not yet understood.
Animal models for Parkinson’s disease have provided some insight into Parkinson’s disease; however, as rodents do not naturally develop Parkinson’s disease, animal studies proved unsatisfactory in recapitulating hallmark features of the disease. In addition, the human brain contains many more dopaminergic neurons, which also wire up differently within the human brain, sending projections to the striatum and the cortex.
“We sought to develop an in vitro model that recapitulates these human features in so-called brain organoids,” explains Daniel Reumann, previously a Ph.D. student in the lab of Jürgen Knoblich at IMBA and first author of the paper. “Brain organoids are human stem cell-derived three-dimensional structures, which can be used to understand both human brain development and function,” he explains further.
The team first developed organoid models of the so-called ventral midbrain, striatum, and cortex—the regions linked by neurons in the dopaminergic system—and then developed a method for fusing these organoids together. As happens in the human brain, the midbrain organoid’s dopaminergic neurons send projections to the striatum and the cortex organoids.
“Somewhat surprisingly, we observed a high level of dopaminergic innervation, as well as synapses forming between dopaminergic neurons and neurons in striatum and cortex,” Reumann recalls.
To assess whether these neurons and synapses are functional, the team collaborated with Cedric Bardy’s group at SAHMRI and Flinders University, Australia, to investigate if neurons in this system would start to form functional neural networks. Indeed, when the researchers stimulated the midbrain, which contains dopaminergic neurons, neurons in the striatum and cortex responded to the stimulation. “We successfully modeled the dopaminergic circuit in vitro, as the cells not only wire correctly, but also function together,” Reumann says.
The organoid model of the dopaminergic system could be used to improve cell therapies for Parkinson’s disease. In first clinical studies, researchers have injected precursors of dopaminergic neurons into the striatum, to try and make up for the lost natural innervation.
However, these studies have had mixed success. In collaboration with the lab of Malin Parmar at Lund University, Sweden, the team demonstrated that dopaminergic progenitor cells injected into the dopaminergic organoid model mature into neurons and extend neuronal projections within the organoid.
“Our organoid system could serve as a platform to test conditions for cell therapies, allowing us to observe how precursor cells behave in a three-dimensional human environment,” Jürgen Knoblich, the study’s corresponding author, explains. “This allows researchers to study how progenitors can be differentiated more efficiently and provides a platform which allows [us] to study how to recruit dopaminergic axons to target regions, all in a high-throughput manner.”
Insights into the reward system
Dopaminergic neurons also fire whenever we feel rewarded, thus forming the basis of the “reward pathway” in our brains. But what happens when dopaminergic signaling is perturbed, such as in addiction? To investigate this question, the researchers made use of a well-known dopamine reuptake inhibitor, cocaine. When the organoids were exposed to cocaine chronically over 80 days, the dopaminergic circuit changed functionally, morphologically, and transcriptionally.
These changes persisted, even when cocaine exposure was stopped 25 days before the end of the experiment, which simulated the withdrawal condition. “Even after almost a month after stopping cocaine exposure, the effects of cocaine on the dopaminergic circuit were still visible, which means that we can now investigate what the long-term effects of dopaminergic overstimulation are in a human-specific in vitro system,” Reumann says.
More information:
Jürgen Knoblich et al, In vitro modeling of the human dopaminergic system using spatially arranged ventral midbrain–striatum–cortex assembloids, Nature Methods (2023). DOI: 10.1038/s41592-023-02080-x
Journal information:
Nature Methods
Source: Read Full Article



